Culturing of human retinal pigment epithelial cells (hRPE) is the initial step in cell therapy of some retinal diseases. To transfer these cells into clinical use, it is necessary to guarantee that they are well differentiated and contamination free. Fluorescence microscopy is the easiest method to do this, but it is associated with operator subjectivity, and the results are highly variable. The aim of this study was to demonstrate the practicality of implementing flow cytometry (FC) analysis to determine the purity of human RPE primary cell cultures. An ARPE19 cell line, human skin fibroblasts, hRPE, and human corneal epithelial cells were analysed by FC to determine the percentage of the hRPE population expressing RPE65 and epithelial and fibroblast proteins. The cell viability and DNA content also were determined. FC analysis showed that the hRPE cells were healthy, stable, and expressed RPE65 protein in the study working conditions. The density of RPE65 protein expression decreased during passages 2 to 10, which was confirmed using a Western blot technique. However, the hRPE cells did not express the 112-kDa epithelial and fibroblast proteins in the current working conditions. These findings suggested that FC facilitates a detailed analysis of human RPE primary cell cultures, a necessary step in developing new cell therapies for retinal diseases.
 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...


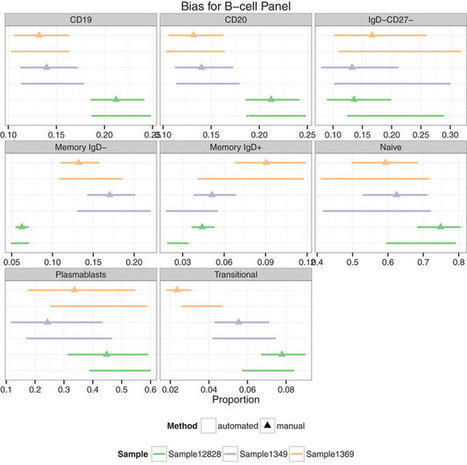




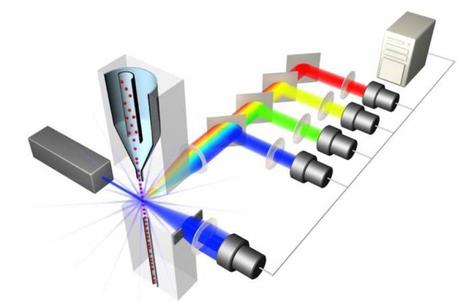

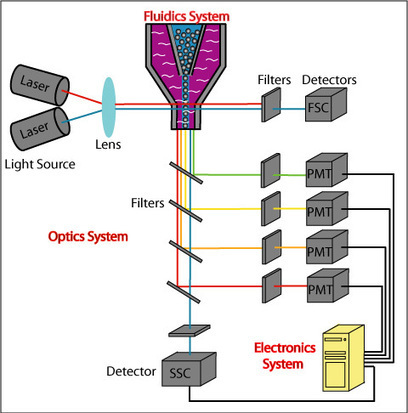

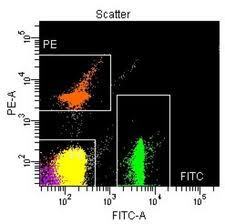
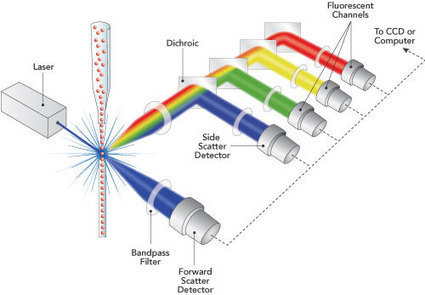


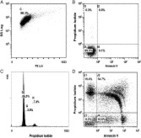









This study follows the “open science” trend by providing complete transparency of data and results, ensuring that reproducibility can be verified21,22,26. All materials, including primary data files, processed data, workspaces and analysis code, are made freely available using existing data standards and providing a valuable resource to the experimental and computational communities.